The patented Safe-2-Dose™ Integrated Dosing Container is the next generation Infants elixir dispenser providing enhanced medication dosing accuracy and user convenience. The caregiver need only disabl

Safe-2-Dose™ Integrated Dosing Container
The patented Safe-2-Dose™ is designed to specifically administer a precise patient calibrated dose of acetaminophen between the U.S. FDA tentative final monograph (TFM), and original safety and efficacy studies for the dosing approval of 4.54mg/lb to 6.80mg/lb (10-15mg/kg). More specifically, the Safe-2-Dose™ is designed to deliver a highly precise dose to a specific patient characteristic within this range. The Safe-2-Dose™ may be calibrated to deliver an optimal mid-point dose from the original safety and efficacy studies of infants and children’s acetaminophen, such as 5.67/mg/lb (12.5mg/kg), among other fixed doses within the recommended range.
The caregiver need only unlock the device and squeeze the bottle to obtain the correct amount of medication to the unique displayed multi-indicia for patient characteristic such as weight, age, temperature, BSA, BMI or drug dose. The Safe-2-Dose’s calibrated indicia enhances safety by overall user convenience and patient dosing accuracy.
The patented Safe-2-Dose™ is the next generation liquid medication dispenser. Each Safe-2-Dose™ is calibrated to the dosage strength of a specific medication and marked with multiple correlated indicia measurements. These measurements may include, among others, weight or other patient characteristic, medication dosage or volume of the medication. The enhanced multi-indicia display increases the overall safety and accuracy of administration, by providing the ability to intuitively correlate the unit dosage to a specific patient characteristic.
The Safe-2-Dose™ eliminates the need to correlate a volume, unit dosage or patient feature from a medication package or other media to the delivery device. These features circumvent the procedures shown to be the primary cause of error during the preparation and administration of OTC liquid medications.4
The Safe-2-Dose™ provides un-matched safety and convenience during preparation and administration, while in parallel, providing ultra-precise dosing and visual reverification of the correct amount of medication for the specific patient.

Features and Benefits:
- Ultra-Precise Dosing Measurements for all recommended Dosing Modes
- Calibrated patient characteristic Indica featuring, weight, age, temperature, BMI, BSA, among others.
- Volumetric Dosing in mL, mg or other Potency
- Child Resistant Control Features
- Calculation and Correlation Free
- Lower More Precise Lower Incremental Indicia
- Snap On-Off Applicator for Easy Washing
- Applicator shipped in Separate PE Pouch
- Gamma Sterilization of Container and Applicator
Infants and Children’s Acetaminophen
The American Academy of Pediatrics, U.S. FDA, and Australian TGA among other health agencies, recommend to dose acetaminophen to a child’s weight, then alternatively to age only if weight is not known.1 Dosage of acetaminophen based on a child weight and/or body surface area, is recognized as the most accurate basis for calculating the optimal drug dosage.2
This optimal weight-based dosing has been validated through dozens of clinical studies. This data has been published since 1976, including by the leading brand producer of acetaminophen in the U.S. by clinical submissions to the FDA in 2009.3
Conversely, no oral delivery applicators included within acetaminophen products have a weight and age indicia.
Studies and statistical data continue to corroborate the leading causes of medication error during the administration of children’s and infants’ acetaminophen. The most common errors stem from an incorrect dose primarily associated from the correlation between the dosing applicator and the medication instructions and/or packaging.4
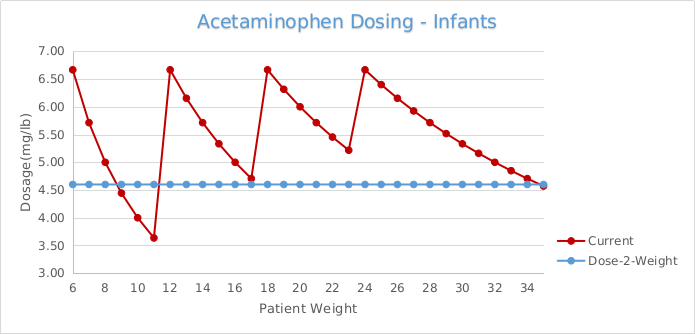
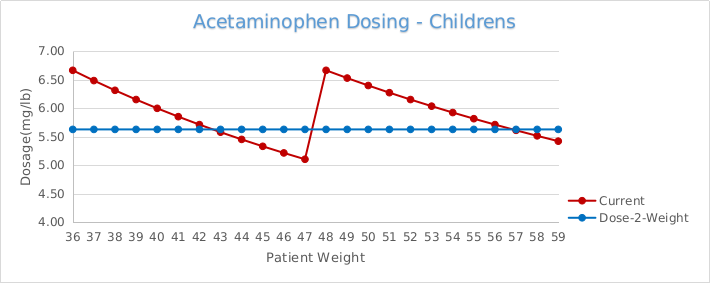
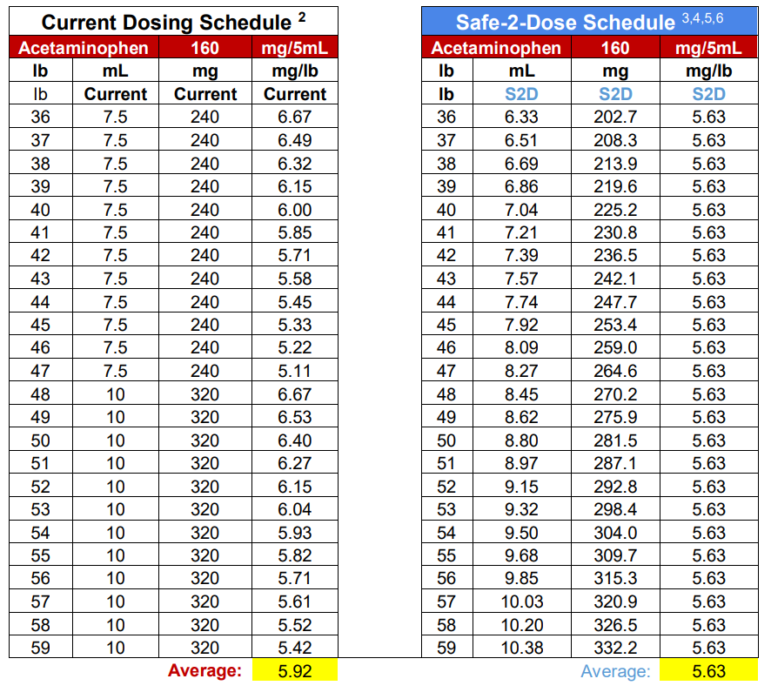
Further exacerbating potential dosing errors, the current U.S mL dosing regimen provides highly variable dosing to sensitive small mass pediatric patients. For example, the current mL dosing provides the lowest weight and youngest patients (24lb at 24 months), the near maximum dose of 6.67 mg/lb (14.70 mg/kg), and the heaviest weight and oldest patient (35 lb at 36 months) in the same range, receiving the lowest possible dose of 4.57mg/lb (10.07 mg/kg).5 More specifically, FDA submissions and original supporting clinical safety and efficacy studies identify the optimal dosing at 12.5mg/kg, and clearly demonstrate the extreme limits provided by this dosing regimen while attempting to control the dose between the 10-15mg/kg range.5
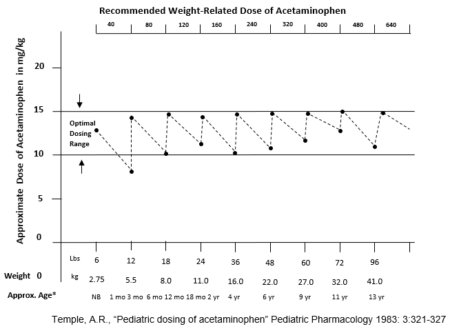
This highly variable dosing continues to be US industry practice, although the original 1983 weight-based dosing schedule published was intended to provide increases in dose corresponding with incremental increases in weight”.6 That study highlighted this weight-based dosing regimen to provide a more consistent mg/kg dosing pattern.6
More importantly, the original safety and efficacy studies published for the approval of infants’ and children’s acetaminophen; clearly demonstrate the minimum effective dose at a fixed unit dose of 10mg/kg (4.54mg/lb), and not as a variable range dose.6,7 In addition, one of the largest pediatric drug trials ever conducted, and regarded pivotal for the pediatric approval of acetaminophen, was completed and its results published utilizing a fixed unit dose of 12mg/kg.8
1] United States Food and Drug Administration. Meeting of the Nonprescription Drugs Advisory Committee. May 17-18, 2011. https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/NonprescriptionDrugsAdvisoryCommittee/UCM264148.pdf
2] Zenk KE,(1979): Drug dosing in children. In Pagliaro LA, Levin RH (eds):“Problems in Pediatric Drug Therapy”. Drug Intelligence Pub, Inc. pp 209-219
3] Rane A. Wilson JT (1976): Clinical pharmacokinetics in infants and children. Clin Pharmacokin 1:2-24
4] Hurwitz J, Sands S, Davis E, Nielsen J, Warholak T. Patient knowledge and use of acetaminophen in over-the-counter medications. J Am Pharm Assoc (2003)
5] Tylenol® Infants and Children’s Dosing Information for Healthcare. https://www.tylenol.com/children-infants/safety/dosage-charts McNeil Consumer Healthcare.
6] FDA Citizen Petition February 1,1999. https://ia600405.us.archive.org/29/items/607305-19 99-mcneil-citizen-petition-on-pediatrics/607305-1999-mcneil- citizen-petition-on-pediatrics.pdf
7] Temple, A.R., “Pediatric dosing of acetaminophen”. Pediatric Pharmacology 1983; 3:321-327
8] Boston University Fever Study. 1994 http://www.bu.edu/slone/research/scor-network/
Infants Safe-2-Dose™ Acetaminophen Oral Dispensers
Researchers have shown that only 30% of caregivers are able to demonstrate both an accurately measured and correct dose for their child.1 Generally over-the-counter (OTC) medications are considered safe, however they are the most frequently implicated pharmaceuticals involved in cases reported to the American Association of Poison Control Center’s National Poison Data System.2
Current dosing recommendations for OTC oral medications commonly specify a volume of medication for a weight or age range, rather than using the dosage strength of the medication specific to an individual patient weight. This dosing regimen results with patients in a designated range receiving the same amount of medication, even though their individual weight may differ more than 45% (6-11lbs); and patients in different ranges receiving double the medication (40-80mg) despite having a difference in weight of 1lb, (11-12lbs).
1] Simon, H. K., & Weinkle, D. A. (1997). Over-the-counter medications: do parents give what they intend to give? Archives of pediatrics & adolescent medicine, 151(7), 654-656.
2] Bronstein AC, Spyker DA, Cantilena LR, Green JL, Rumack BH, Heard SE. 2007 annual report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 25th Annual Report. Clin Toxicol (Phila). 2008; 46(10):927-1057.
INFANTS ACETAMINOPHEN
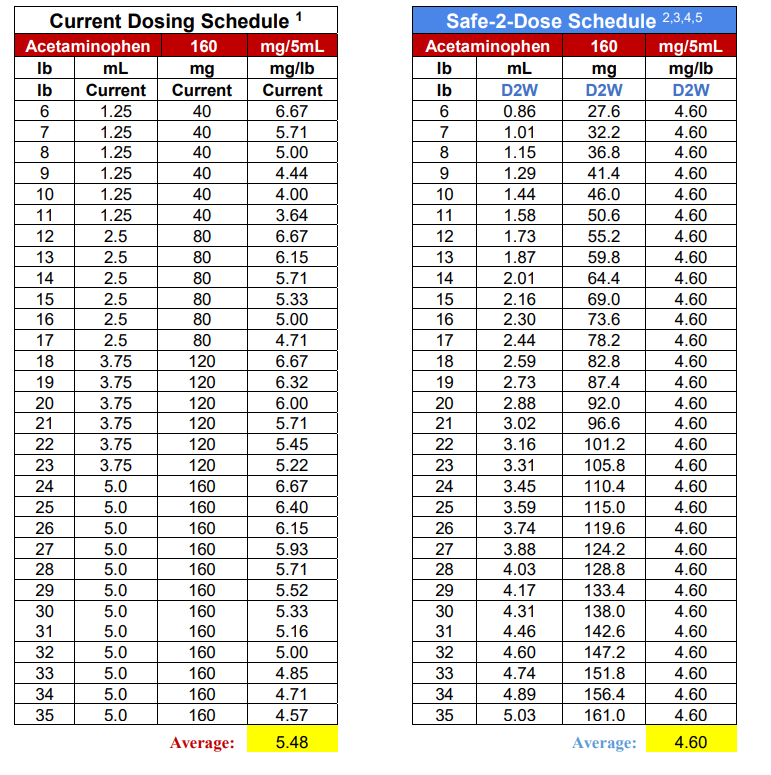
1] Tylenol® Infants and Children’s Dosing Information for Healthcare Professionals
2] Adjusted for U.S. Market and mL dosing conformity – (Safe-2-Dose™ Optimal Dose of 12.5mg/kg (5.67 mg/lb.) to 10.14mg/kg (4.60 mg/lb.))
3] US Food and Drug Administration – Over the counter Acetaminophen –Containing Drug Products in Children Background Package – Efficacy and Safety Studies published and reviewed at 4.54 mg/lb. to 6.80 mg/lb. Publish Studies mid-point and Safe-2-Dose™ Optimal Recommended Dose 5.67 mg/lb.
4] American Academy of Pediatric Endorses Weight Based Dosing – Recommended Dosing at 4.54 mg/lb. to 6.80 mg/lb. mid-point and Safe-2-Dose™ Optimal Recommended Dose 5.67 mg/lb.
Tylenol® is a registered trademark of Johnson & Johnson.
Children's Safe-2-Dose™ Acetaminophen Oral Dispensers

To promote dosing accuracy, both the American Academy of Pediatrics (AAP) and the US Food and Drug Administration (FDA) recommend that parents use dosing tools that are paired with the specific medication.
Oral syringes are considered the gold standard when accuracy is critical.1 Nevertheless, Dosing cups are the most frequently included dosing devices included with OTC medications.
Studies have shown a >4 times increase of error when dosing cups are use verses syringes.1
Nevertheless, due to the increase in volume of the medication required for Children verses Infants; as well as perhaps user preference, syringes are mainly utilized for infants’ administration.
The Safe-2-Dose™ is designed to provide the safety and accuracy of an oral syringe while offering the convenience of a spoon or dosing cup. The Safe-2-Dose™ is calibrated to the dosage strength and marked with multiple correlated indicia measurements. These measurements may include, among others, weight or other patient characteristic, medication dosage or volume of the medication. The enhanced multi-indicia display increases the overall safety and accuracy of administration, by providing the ability to intuitively correlate the unit dosage to a specific patient characteristic.
CHILDREN’S ACETAMINOPHEN
1] Yin HS, Parker RM,Sanders LM, et al. Liquid Medication Errors and Dosing Tools: A Randomized Control Experiment. Pediatrics 2016;138(4):e20160357
2] Tylenol® Infants and Children’s Dosing Information for Healthcare Professionals
3] Adjusted for U.S. Market and mL dosing conformity – (Safe-2-Dose™ Optimal Dose of 12.5mg/kg (5.67 mg/lb.) to 12.41mg/kg (5.63 mg/lb.))
4] US Food and Drug Administration – Over the counter Acetaminophen –Containing Drug Products in Children Background Package – Efficacy and Safety Studies published and reviewed at 4.54 mg/lb. to 6.80 mg/lb. Publish Studies mid-point and Safe-2-Dose™ Optimal Recommended Dose 5.67 mg/lb.
5] American Academy of Pediatric Endorses Weight Based Dosing – Recommended Dosing at 4.53 mg/lb. to 6.80 mg/lb. mid-point and Safe-2-Dose™ Optimal Recommended Dose 5.67 mg/lb.
Tylenol® is a registered trademark of Johnson & Johnson.
The Safe-2-Dose® is a registered trademark of Asepsis Medical Technologies, LLC., and covered by one or more patents, including; US Patent 11,517,505 and Patents Pending.
Latest News
Asepsis Announces The Grant Of Mexico Patent No. 384,129
Asepsis Medical Technologies® is proud to introduce the latest addition to the Calibrated Drug Delivery™ (CCD) platform, the Multi-Dose Safe-2-Dose® syringe. The Multi-Dose Safe-2-
Asepsis Announces the Grant of Canadian Patent No. 3,006,218
Asepsis Medical Technologies® is proud to announce their latest Canadian Patent Grant 3,006,218 of August 24, 2021 – for the multi-dose oral Dose-2-Weight Syringe®. The Dose-2-Weight Sy
Asepsis announces the US Patent Grant 11,058,606
Asepsis Medical Technologies announces the USPTO issuance of Patent Application Number 11,058,606 for its Safe-T-Fuse™ drug infusion system. The Safe-T-Fuse™ is an industry unique infusion contai
Asepsis Announces the Grant of Mexico Patent MX/a/2017/012034
The Safe-T-Syringe® is designed to deliver the highest standard of patient care via syringe. Its medication calibrated indicia provide enhanced accuracy, convenience and safety through-out preparation
Asepsis Announces the Grant of U.S. Patent 10,857,299
Asepsis Medical Technologies® is proud to introduce the latest addition to the Calibrated Drug Delivery™ (CCD) platform, the Multi-Dose Safe-2-Dose® syringe. The Multi-Dose Safe-2-Dose® syringe provid
Asepsis Announces the U.S. Patent Grant No. 10,625,026
Asepsis Medical Technologies announces the U.S. Patent Grant for the Momenta-Dose™ forcé actuated injection device. The Momenta Dose™ is designed to administer rapid small volume subcutaneous or intra


